MILWAUKEE — Two new COVID-19 antiviral pills are being dubbed potential game-changers in the effort to curb coronavirus hospitalizations and deaths.
Medical experts say these COVID pills that are currently up for FDA approval are not a substitute for vaccinations. While there are high hopes these pills could minimize hospitalizations, trials show one pill is outperforming its competitor.
Nearly two years into the pandemic, drugmakers Pfizer and Merck say they both have a pill ready to be manufactured to prevent serious COVID illness and death.
To better understand both drugs that may be available at pharmacies in just a couple months, let’s go ‘360’. An emergency medical physician explains how the pills work, a Milwaukee woman who’s unvaccinated sheds light on possible hesitancy, and a St. Francis man shares why he hopes the pills could keep others from what he went through.
"It's not an experience that anybody should go through. It's harrowing, it's difficult,” Chris Stolarski said. “There were days where I could easily have died."
Stolarski says he spent more than two weeks in an intensive care unit with COVID last November. At the time in 2020, vaccines were unavailable and therapeutics were much more limited.
Stolarski says his battle with lingering COVID didn’t stop when he was discharged from the hospital. He had to be on oxygen for another month once he got home.
"You don't want to get to that point, but this is a great back-up parachute that we have, and I'm particularly encouraged for people who are immune-deficient in some way who can't have the vaccine or for whom the vaccine isn't particularly effective,” he said.
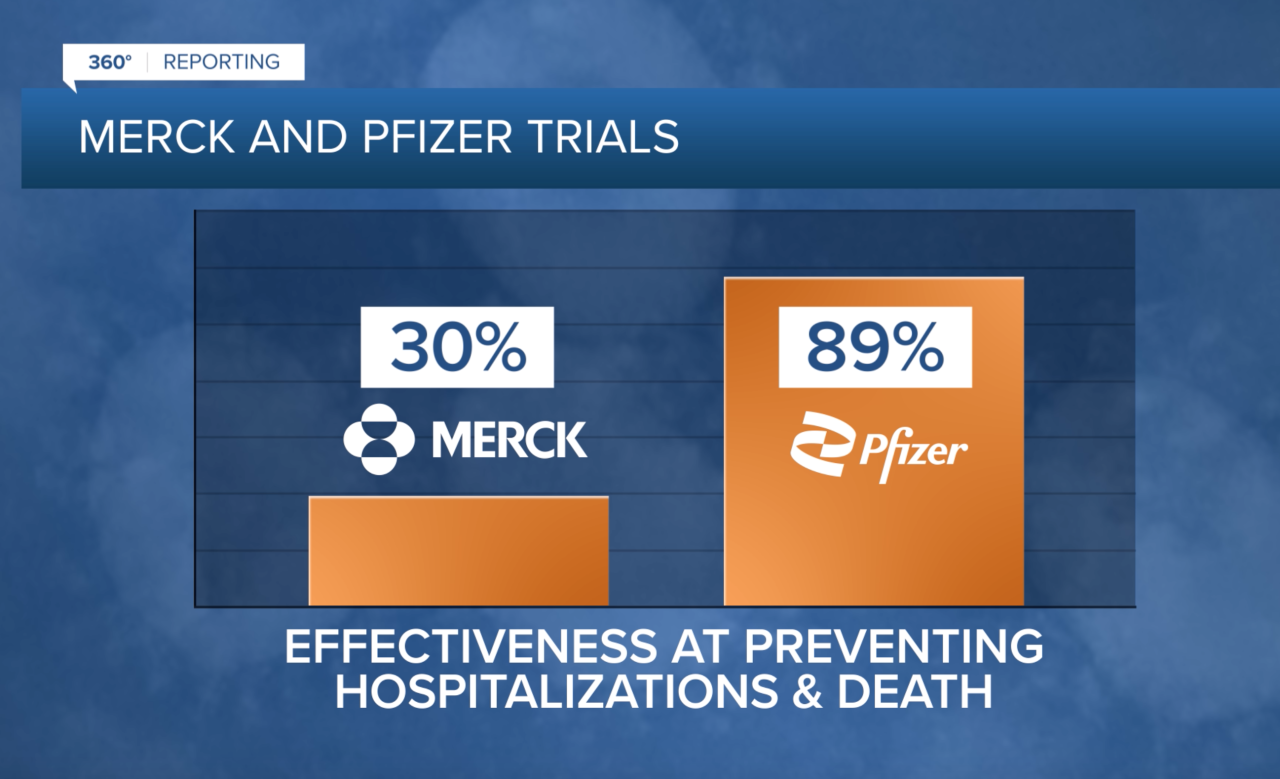
Both pills are designed to be taken for five days after COVID symptoms appear. Merck’s pill was initially developed nearly two decades ago to treat the flu. The company pivoted during the pandemic and recent trials found its pill to be 30 percent effective at preventing coronavirus hospitalizations and deaths.
Pfizer says trials found its brand new COVID pill to be 89 percent effective.
"This is something that's new, it's in the experimental stage, I'm not willing to be guinea pig,” Sarah Scott of Milwaukee said.
Scott said she is unvaccinated and wouldn’t take antiviral pills due to concerns over potential long-term effects. Merck says its pill should not be used by kids or pregnant women due to the possible risk of birth defects among newborns.
"I don't want to take no pill, I don't want to take no vaccination, I'm going to go home and start building my immune system more and more which I do every day,” Scott said.
- The Rebound Milwaukee: Resources For Getting Back To Normal
- We're Open: These Restaurants Are Still Offering Carryout And Delivery
Dr. Jeff Pothoff with UW Health says the best defense against the virus is being fully vaccinated and he believes that will continue to be the case if and when these COVID antiviral pills hit the market.
"Being realist and realizing there are a number of folks here in the United States and around the world that are not going to be vaccinated, having something that prevents them or reduces the likelihood that they go onto severe disease, being on a ventilator, overwhelming hospitals, dying, that's where the game changer is,” he said.
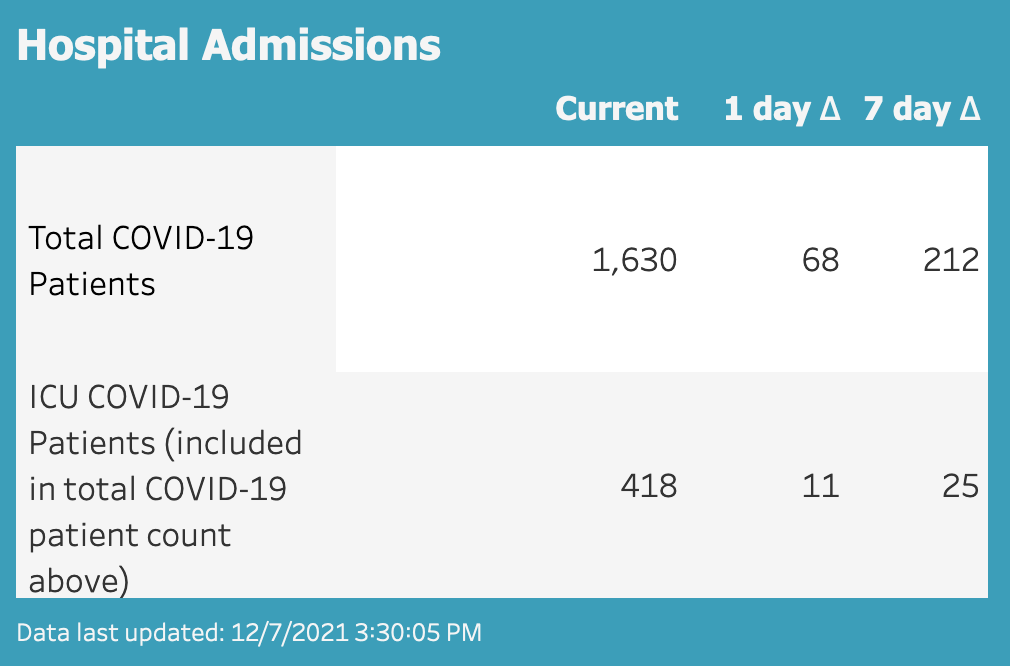
The Wisconsin Hospital Association shows COVID hospitalization rates are the highest they’ve been in a year with more than 1,600 coronavirus patients. 418 of whom are reportedly in intensive care units.
Dr. Pothoff says Merck and Pfizer’s pills both use a similar technology to stop the virus from replicating and becoming worse once it’s inside the body.
"I think the real benefit of these antivirals is really in the delivery. It's easy to make pills, stock pills, they won't expire as much, it's not as much work,” Dr. Pothoff said.
The Food and Drug Administration is expected to make its final decision on Merck’s pill by the end of December. Pfizer is next in line and is expecting an FDA decision in January.




